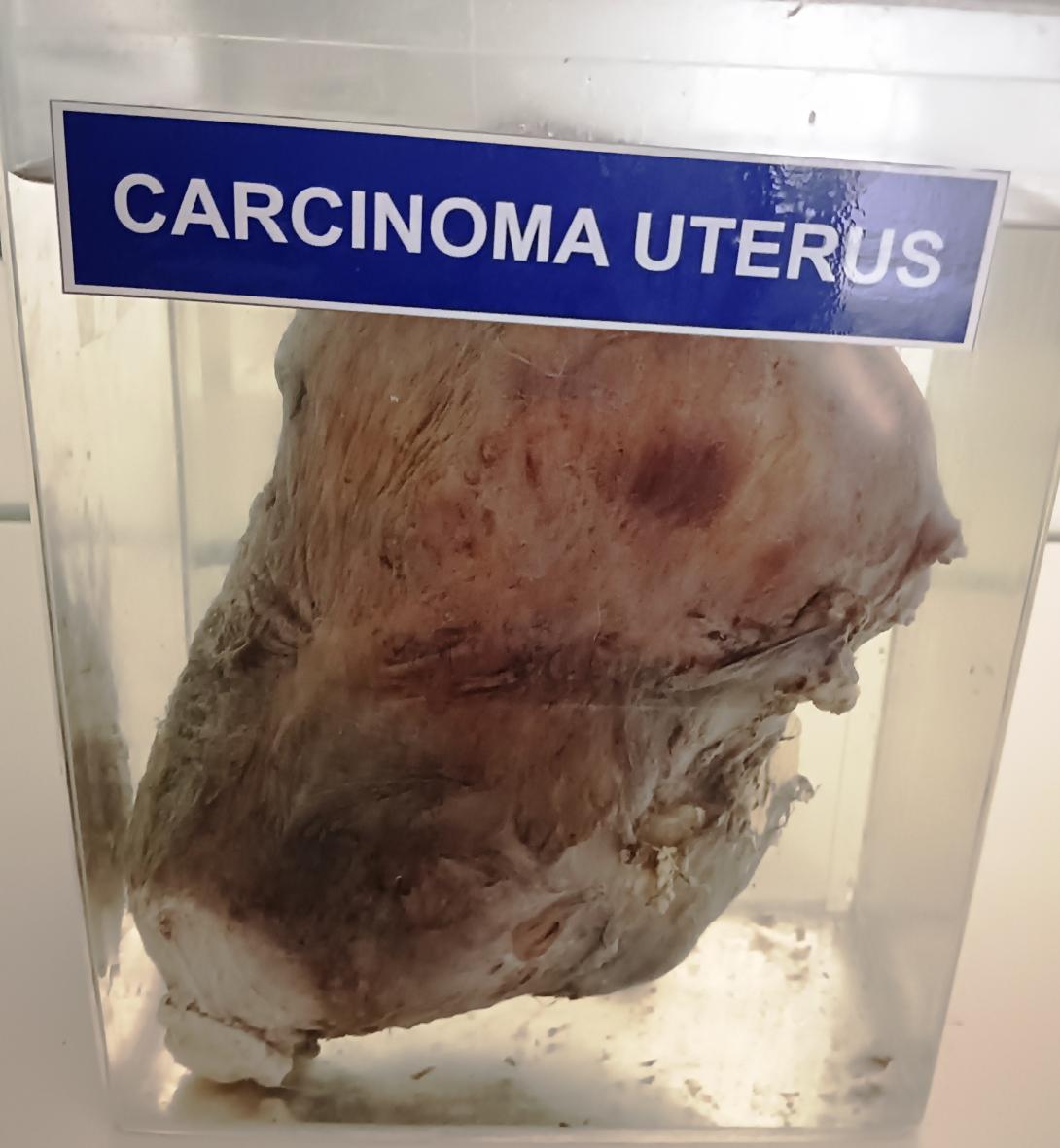
Carcinoma of the uterus is a type of cancer that begins in the cells of the uterus, which is a hollow, muscular organ located in the pelvis of females. The gross anatomy of carcinoma of the uterus can vary depending on the stage of the disease, but here are some general features:
- Enlargement of the uterus: The uterus may be larger than normal due to the presence of a tumor. The tumor can cause the uterus to grow in size and weight.
- Irregular shape: The uterus may have an irregular shape due to the presence of the tumor. The tumor can cause the uterus to bulge in certain areas, making it appear irregular.
- Abnormal growths: The tumor may appear as a mass or growth on the surface of the uterus. It may also invade the wall of the uterus, causing the tissue to thicken and become irregular.
- Tissue changes: The tissue of the uterus may become pale, soft, and friable (easily crumbled) due to the cancer.
- Bleeding: Carcinoma of the uterus can cause abnormal vaginal bleeding, which may include heavy bleeding, bleeding between periods, or bleeding after menopause.
- Lymph node involvement: In advanced stages, the cancer may spread to nearby lymph nodes, causing them to become enlarged and palpable.
It is important to note that these gross anatomical features may vary depending on the specific type of carcinoma of the uterus and the stage of the disease. Diagnosis and treatment should always be carried out by a medical professional.
Rack Number
Specimen Number
56